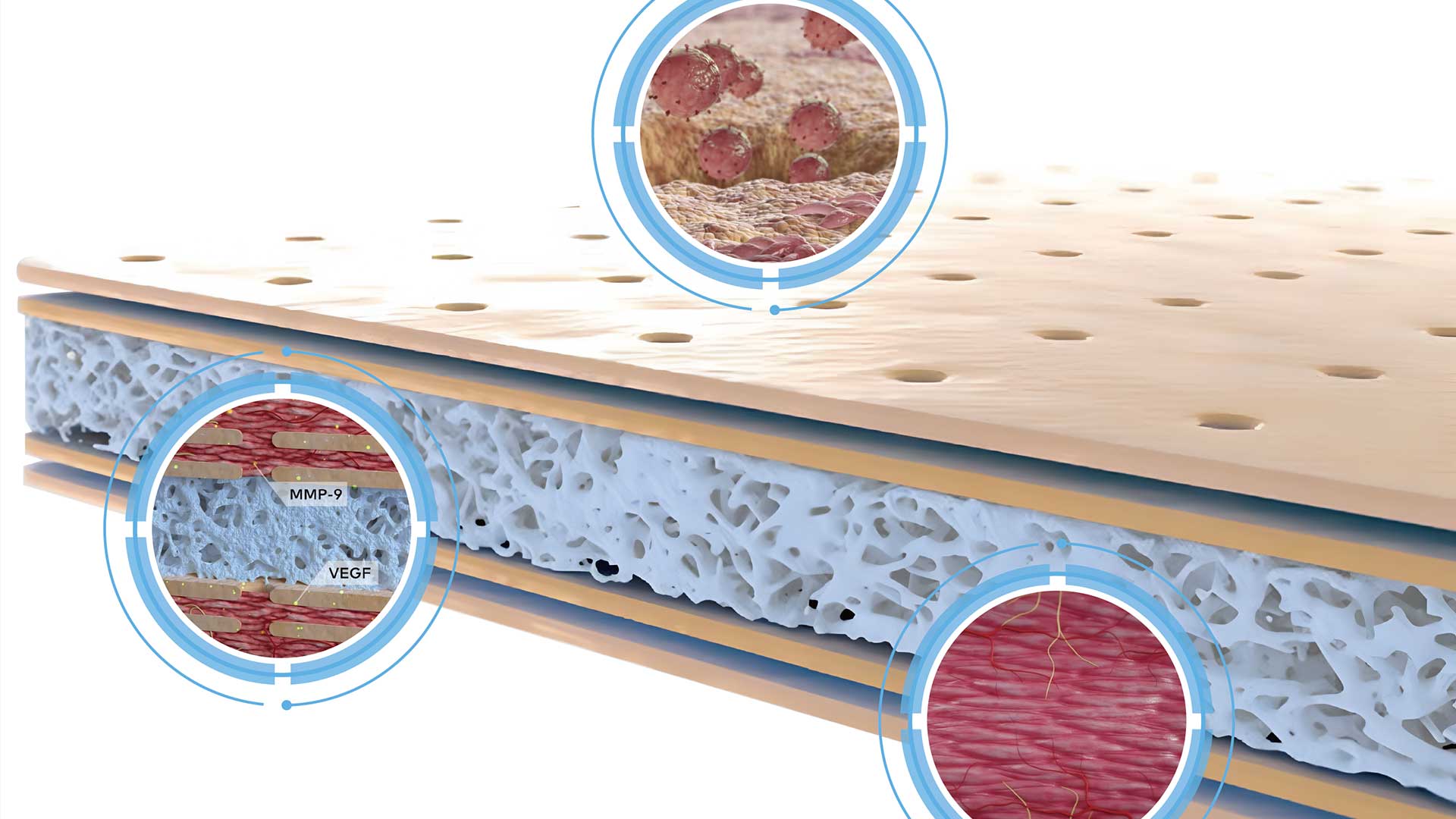
ZR MedTech
The Team

Yanyan Wang
-
Chairman of the Board and Chief Executive Officer
ZR MedTech
Innovation to Regeneration
We deliver innovative excellence for surgeons to regenerate patients’ life.
15 years ago, I was challenged by a surgeon that there was no real long-term solution for treating tissue defects. Many patients suffering from recurrence or no effective clinical solutions for biodegraded tissue restoration postoperatively. In these years, we continued to develop our technology encouraged by our friends and partners from clinic and scientific works. We learned to understand deeper of biology and respect of it. Subsequent iteration of our technology was never been paused. Now, our technology is being able to help millions of people recovering from diseases, and we are still on the way of understanding unknown, and providing new solutions for the same question.
We deliver innovative excellence for surgeons to regenerate patients’ life.
We only bring product excellence.
— Dr. Yanyan Wang,
Founder of ZR Medtech
Our
History
Our
Mission

Leader of Surgical Tissue Reconstruction and Critical Management
Innovation to
Regeneration
-
ZR MedTech Founded in 2015
-
Currently own 5 PCT Patents、2 NMPA Class lll Medical Device Registration Certificates、2 “Innovative Green Pathway for Medical Devices” by NMPA
-
30+ pipeline supported by strong R&D capacity
-
Intelligent Manufactory with ISO13485 & MDSAP
5500m2
Taicang
2200m2
Shanghai R&D Center
100000m2
Shanghai One-Site Center (2025)